Page 182 - Kỷ yếu hội thảo quốc tế: Ứng dụng công nghệ mới trong công trình xanh - lần thứ 9 (ATiGB 2024)
P. 182
th
HỘI THẢO QUỐC TẾ ATiGB LẦN THỨ CHÍN - The 9 ATiGB 2024 173
−
4 Zn 2+ + SO ²− + 6 OH + 3 H O
4 2
(1)
→ Zn 4SO OH 3 H 0
( ) •
4
6 2
4. CONCLUSIONS
Summarily, the resistance to dendrite formation in
zinc ion pouch cells was evaluated over 50 hours at a
2
current density of 1.0 mA/cm using electrolytes of
2M ZnSO4 with varying percentages of DMSO (0%,
10%, 20%, 30%, 40%, and 50%). The zinc metal
electrode surfaces were examined after 50 hours of
charge - discharge cycling in a symmetric pouch cell
configuration. The formation of complex layers on the
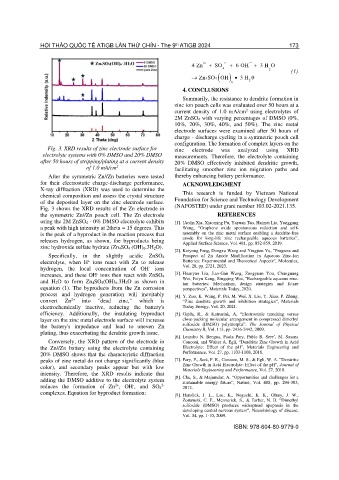
Fig. 3. XRD results of zinc electrode surface for zinc electrode was analyzed using XRD
electrolyte systems with 0% DMSO and 20% DMSO measurements. Therefore, the electrolyte containing
after 50 hours of stripping/plating at a current density 20% DMSO effectively inhibited dendritic growth,
of 1.0 mA/cm 2 facilitating smoother zinc ion migration paths and
After the symmetric Zn//Zn batteries were tested thereby enhancing battery performance.
for their electrostatic charge-discharge performance, ACKNOWLEDGMENT
X-ray diffraction (XRD) was used to determine the
chemical composition and assess the crystal structure This research is funded by Vietnam National
of the deposited layer on the zinc electrode surface. Foundation for Science and Technology Development
Fig. 3 shows the XRD results of the Zn electrode in (NAFOSTED) under grant number 103.02-2021.135.
the symmetric Zn//Zn pouch cell. The Zn electrode REFERENCES
using the 2M ZnSO4 - 0% DMSO electrolyte exhibits [1]. [Aolin Xia, Xiaoming Pu, Yayuan Tao, Haimei Liu, Yonggang
a peak with high intensity at 2theta = 15 degrees. This Wang, “Graphene oxide spontaneous reduction and self-
is the peak of a byproduct in the reaction process that assembly on the zinc metal surface enabling a dendrite-free
releases hydrogen, as shown, the byproducts being anode for long-life zinc rechargeable aqueous batteries”,
Applied Surface Science, Vol. 481, pp. 852-859, 2019.
zinc hydroxide sulfate hydrate (Zn 4SO4 (OH)6.3H2O).
[2]. Kaiyong Feng, Dongxu Wang and Yingjian Yu, “Progress and
Specifically, in the slightly acidic ZnSO4 Prospect of Zn Anode Modification in Aqueous Zinc-Ion
electrolyte, when H ions react with Zn to release Batteries: Experimental and Theoretical Aspects”, Molecules,
+
-
hydrogen, the local concentration of OH ions Vol. 28, pp. 2721, 2023.
-
increases, and these OH ions then react with ZnSO 4 [3]. Huanyan Liu, Jian-Gan Wang, Zongyuan You, Chunguang
and H2O to form Zn 4SO4(OH)6.3H2O as shown in Wei, Feiyu Kang, Bingqing Wei, “Rechargeable aqueous zinc-
ion batteries: Mechanism, design strategies and future
equation (1). The byproducts from the Zn corrosion perspectives”, Materials Today, 2020.
process and hydrogen generation will inevitably [4]. Y. Zuo, K. Wang, P. Pei, M. Wei, X. Liu, Y. Xiao, P. Zhang,
convert Zn 2+ into "dead zinc," which is “Zinc dendrite growth and inhibition strategies”, Materials
electrochemically inactive, reducing the battery's Today Energy, Vol. 20, 2021.
efficiency. Additionally, the insulating byproduct [5]. Gajda, R., & Katrusiak, A, “Electrostatic matching versus
layer on the zinc metal electrode surface will increase close-packing molecular arrangement in compressed dimethyl
the battery's impedance and lead to uneven Zn sulfoxide (DMSO) polymorphs”. The Journal of Physical
plating, thus exacerbating the dendrite growth issue. Chemistry B, Vol. 113, pp. 2436-2442, 2009.
[6]. Leandro N. Bengoa, Paola Pary, Pablo R. Sere´, M. Susana
Conversely, the XRD pattern of the electrode in Conconi, and Walter A. Egli, “Dendritic Zinc Growth in Acid
the Zn//Zn battery using the electrolyte containing Electrolyte: Effect of the pH”, Materials Engineering and
20% DMSO shows that the characteristic diffraction Performance, Vol. 27, pp. 1103-1108, 2018.
peaks of zinc metal do not change significantly (blue [7]. Pary, P., Seré, P. R., Conconi, M. S., & Egli, W. A. “Dendritic
color), and secondary peaks appear but with low Zinc Growth in Acid Electrolyte: Effect of the pH”, Journal of
Materials Engineering and Performance, Vol. 27, 2018.
intensity. Therefore, the XRD results indicate that
adding the DMSO additive to the electrolyte system [8]. Chu, S., & Majumdar, A. “Opportunities and challenges for a
sustainable energy future”, Nature, Vol. 488, pp. 294-303,
reduces the formation of Zn , OH , and SO4 2012.
-
2+
2-
complexes. Equation for byproduct formation: [9]. Hanslick, J. L., Lau, K., Noguchi, K. K., Olney, J. W.,
Zorumski, C. F., Mennerick, S., & Farber, N. B. “Dimethyl
sulfoxide (DMSO) produces widespread apoptosis in the
developing central nervous system”, Neurobiology of disease,
Vol. 34, pp. 1-10, 2009.
ISBN: 978-604-80-9779-0