Page 167 - Kỷ yếu hội thảo quốc tế: Ứng dụng công nghệ mới trong công trình xanh - lần thứ 9 (ATiGB 2024)
P. 167
158 TRƯỜNG ĐẠI HỌC SƯ PHẠM KỸ THUẬT - ĐẠI HỌC ĐÀ NẴNG
The Zn/LFP pouch cell is assembled using zinc attributed to capacitive-controlled mechanisms at sweep
foil and carbon cloth combined with LiFePO 4. The rates of 0.1, 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 1.0, 1.5 and
cyclic voltammetry (CV) curves for the Zn/LFP 2.5 mV/s, respectively (Fig. 3d).
battery, recorded at a scan rate of 0.1 mV/s, exhibit a
pair of redox peaks within the potential range of 0.8
to 1.6 V (Fig. 3a). This redox peak pair is observed at
1.12 V/1.35 V under the same scan rate,
corresponding to the following reaction:
Cathode: LiFePO4 →Li +e +FePO4 (1)
+
-
2+
-
Anode: Zn + 2e → Zn (2)
The total area under the curve encompasses both
Faradaic and non-Faradaic charge storage mechanisms,
including double-layer capacitive effects. The Faradaic
process itself can be further categorized into two distinct
components: (i) the insertion of Li ions and (ii) charge
transfer linked to surface particles, which is referred to as
the pseudo-capacitance effect. The term 'capacitive'
encompasses all surface charge storage mechanisms,
such as double-layer and pseudo-capacitance effects. To
estimate the “capacitive” behavior, one can analyze the
power law connection between current (i) and scan rates
(v) and calculate the 'b-value' from the linear slope of the
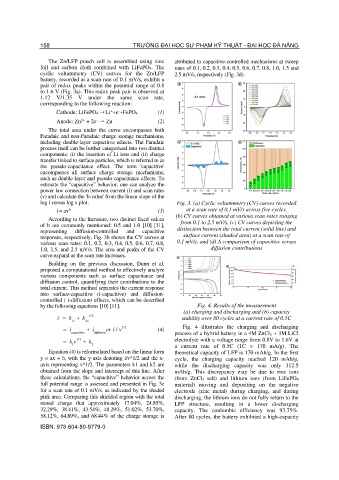
log i versus log v plot: Fig. 3. (a) Cyclic voltammetry (CV) curves recorded
i = av (3) at a scan rate of 0.1 mV/s across five cycles,
b
(b) CV curves obtained at various scan rates ranging
According to the literature, two distinct fixed values
of b are commonly mentioned: 0.5 and 1.0 [10] [11], from 0.1 to 2.5 mV/s, (c) CV curves depicting the
representing diffusion-controlled and capacitive distinction between the total current (solid line) and
responses, respectively. Fig. 3b shows the CV curves at surface current (shaded area) at a scan rate of
various scan rates: 0.1, 0.2, 0.3, 0.4, 0.5, 0.6, 0.7, 0.8, 0.1 mV/s, and (d) A comparison of capacitive versus
1.0, 1.5, and 2.5 mV/s. The area and peaks of the CV diffusion contributions
curve expand as the scan rate increases.
Building on the previous discussion, Dunn et al.
proposed a computational method to effectively analyze
various components such as surface capacitance and
diffusion control, quantifying their contributions to the
total current. This method separates the current response
into surface-capacitive (i-capacitive) and diffusion-
controlled ( i-diffusion) effects, which can be described
by the following equations [10] [11]: Fig. 4. Results of the measurement
(a) charging and discharging and (b) capacity
i = k + k 1/2 stability over 80 cycles at a current rate of 0.5C
1v 2v
= i + i or /i v 1/2 (4) Fig. 4 illustrates the charging and discharging
capacitive diffusion process of a hybrid battery in a 4M ZnCl 2 + 3M LiCl
= k v 1/2 + k electrolyte with a voltage range from 0.8V to 1.6V at
1 2 a current rate of 0.5C (1C = 170 mA/g). The
Equation (4) is reformulated based on the linear form theoretical capacity of LFP is 170 mAh/g. In the first
y = ax + b, with the y-axis denoting i/v^1/2 and the x- cycle, the charging capacity reached 120 mAh/g,
axis representing v^1/2. The parameters k1 and k2 are while the discharging capacity was only 112.5
obtained from the slope and intercept of this line. After mAh/g. This discrepancy may be due to zinc ions
these calculations, the “capacitive” behavior across the (from ZnCl2 salt) and lithium ions (from LiFePO 4
full potential range is assessed and presented in Fig. 3c material) moving and depositing on the negative
for a scan rate of 0.1 mV/s, as indicated by the shaded electrode (zinc metal) during charging, and during
pink area. Comparing this shielded region with the total discharging, the lithium ions do not fully return to the
stored charge that approximately 17.04%, 24.85%, LFP structure, resulting in a lower discharging
32.29%, 38.41%, 43.54%, 48.29%, 51.02%, 53.70%, capacity. The coulombic efficiency was 93.75%.
58.12%, 64.89%, and 68.44% of the charge storage is After 80 cycles, the battery exhibited a high-capacity
ISBN: 978-604-80-9779-0